Results for 'tumor vs. immune system'

Leadership Lab: 4 Ways Biopharma Leaders Can Prepare for Media Interviews
Aug 20th • 6 mins read





MSL Hiring and Recruitment: 5 Ways to Support Diversity and Inclusion
May 19th • 2 mins read



Combatting the “Summer Slow Down" – MSL Job Search Tips for Slower Months
Jun 23rd • 2 mins read



Evaluation of Trials Comparing Single-Enantiomer Drugs to Their Racemic Precursors: A Systematic Review
May 6th • 18 mins read

Updated estimates of eligibility for and response to genome-targeted oncology drugs among US cancer patients, 2006-2020
Apr 20th • 7 mins read
Comparative study on anticancer drug access times between FDA, EMA and the French temporary authorisation for use program over 13 years
Apr 7th • 12 mins read

Virtual Clinical Trials in Oncology-Overview, Challenges, Policy Considerations, and Future Directions
Apr 8th • 4 mins read
Advances in basic research in oncology in 2020: Bridging basic science and clinical care
Mar 24th • 1 min read

Are Quality of Randomized Clinical Trials and ESMO-Magnitude of Clinical Benefit Scale Two Sides of the Same Coin, to Grade Recommendations for Drug Approval?
Feb 11th • 3 mins read

Assessment of Coverage in England of Cancer Drugs Qualifying for US Food and Drug Administration Accelerated Approval
Feb 22nd • 10 mins read

Assessment of Food and Drug Administration- and European Medicines Agency-Approved Systemic Oncology Therapies and Clinically Meaningful Improvements in Quality of Life: A Systematic Review
Feb 11th • 4 mins read
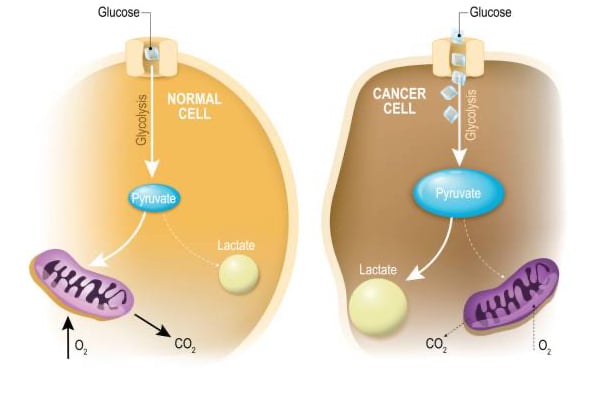
“Oncometabolism: The switchboard of cancer: An editorial”
Feb 1st • 1 min read
